When thinking about biomarkers, we immediately think about blood and urine markers, which involve technology to analyze. But there are many other types of biomarkers that don’t involve taking tissue samples that rely on different types of technology.
Medical imaging is one of them. And it’s important to recognize that the work we do with imaging as a biomarker is not much different than deriving information from a blood sample.
Here, Calyx’s Dr. Peter Steiger reviews current regulations and factors to consider when deciding between investigative site reads and BICR for clinical trial imaging.
What the Regulators Say About Imaging Biomarkers
In 2018, the FDA issued a guidance for industry that remains the global standard for imaging in clinical trials. It was developed with considerable feedback from the industry, including pharmaceutical companies and imaging core labs.
Until recently, there were no direct equivalents to this guidance from other important regulatory agencies like the EMA or the PMDA. But over the past year, the Chinese NMPA has issued two therapeutically focused guidances that are compatible with the FDA guidance; One for oncology, and one for osteoporosis.

“Deciding between BICR and Investigator reads requires careful consideration, which is why it’s very difficult to adjudicate and identify truth in clinical trial imaging.”
– Peter Steiger, PhD, Chief Scientific Officer, Calyx
BICR vs. Investigator Reads: Factors to Consider When Using Imaging Biomarkers
An important component of imaging in clinical trials is the concept of using Blinded Independent Central Review (BICR) which are typically available through central core labs. Although BICR isn’t mandatory, the FDA guidance explains why it is recommended for many trials.
The expectation from the regulatory agencies is that BICR be used in pivotal, later phase trials as it is a more robust way of assessing treatment effects, because it eliminates investigator bias. BICR can sometimes also be used for confirmation of eligibility and progression.
With BICR, readers are trained on study-specific read criteria that are carefully documented in a charter, and only readers who have been trained and monitored for their read quality can read in that trial.
Investigator review, on the other hand, is often sufficient in early phase studies and exploratory work. The issue with investigator review is that it can be influenced by investigator knowledge of clinical data.
But this is not necessarily a concern. In fact, you want the investigator to have all possible information available to assess the patient’s whole health status. For example, when an investigator needs to decide whether to keep a patient on a trial or to take them off experimental treatment, a single investigator read will play an important role. But that can sometimes distract from isolating the signal that is induced by the treatment.
It’s a careful consideration to decide when and where you use either one of these approaches, which is why it’s very difficult to adjudicate and identify truth in clinical trial imaging. And there can be a considerable rate of disagreement between readers, depending on the study indication and how the read criteria are designed. Clinical trial sponsors should be mindful of the fact that there can be disagreement between readers, and plan for that in each trial strategy.
Safety vs. Efficacy
Another important consideration is how the imaging biomarker will be used ‒ to evaluate safety or efficacy. In safety situations, you often need real-time image review and additional clinical data to ensure patient safety can be appropriately assessed. Because the primary objective is the identification of safety signals, the stance is usually aggressive.
In contrast, when evaluating efficacy, the image read isn’t always required for patient management. Therefore, image reviews are typically scheduled in batches, aligned with study milestones and data locks. And the process for managing efficacy reads involves robust management of image quality and controlled access to clinical data.
So, when the primary objective is the identification of efficacy signals, we typically take a more conservative stance.
Complexity
It’s worth considering two principle dimensions of imaging biomarkers in the clinical trial continuum: the time dimension as a drug moves from early phase studies through to approval and post-approval and the different characteristics of a trial’s design.
The purpose of using imaging biomarkers in early phase trials is usually more for exploratory assessment of efficacy. However, once you advance to pivotal trials, the imaging biomarker will usually be well-established to serve as a primary endpoint.
As a result, the tolerance for complexity is high in early-phase studies, while late phase and pivotal studies used for regulatory submission require tight regulatory and scientific control with broadly accepted imaging biomarkers.
Early vs. Late Phase
Often in early phase studies, when you’re making go/no-go decisions, you’re less concerned with regulatory control. It’s perfectly acceptable to use a complex emerging imaging biomarker that is being developed at a leading academic institution during this phase.
Whereas in later phase studies, regulatory validation and approval become critical factors, so less established biomarkers may not suffice. This also translates into the trial’s size and geographical reach, where you go from small and local to large and global.
Breakthrough Designation Changes Everything
We all strive to meet unmet medical needs in a meaningful way and receiving accelerated approval designation in this context is critical. However, for many this journey begins with very limited information. And often as you head into a phase I trial, you’re not exactly sure whether the efficacy signals will warrant an accelerated designation.
This was evident in a recent study where the need for medical imaging escalated when breakthrough therapy designation was granted for a novel oncology compound (see figure) and the simple phase I trial design converted to an advanced phase 3 trial practically overnight.
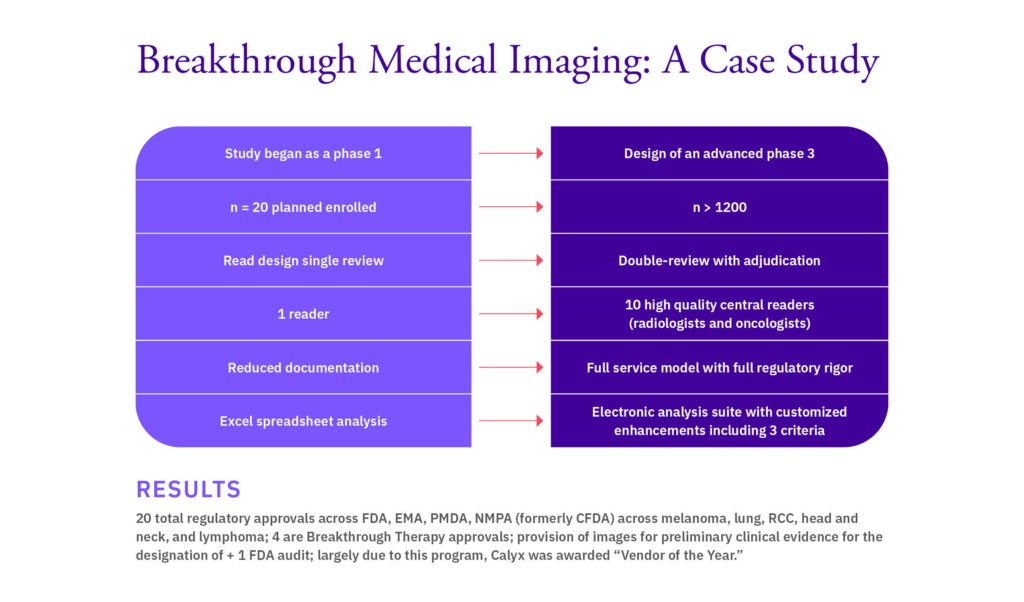
If you have a drug that addresses a very specific unmet medical need, breakthrough therapy designation is a real possibility. And building in flexibility and speaking to your collaborators about what is needed is critical to accelerating your path to approval. You need the right documentation for the regulatory agencies, and you need to have scalable models that can help you get to approval.
Conclusion
Increasingly, getting regulatory approval is not enough to produce a successful drug in the marketplace. We need to demonstrate effectiveness by demonstrating a real-world clinical benefit of the drug. And in demonstrating that, imaging and other biomarkers can play a very important role.
















