Expertise You Can Rely on
By working with Calyx, you have access to experienced scientists and medical professionals who are experts in MSK imaging endpoints and criteria and are up to date on current, global regulations. They deliver technical expertise in the imaging modalities most often used in MSK trials, in protocol design, and in standardizing image acquisition across global investigative sites.
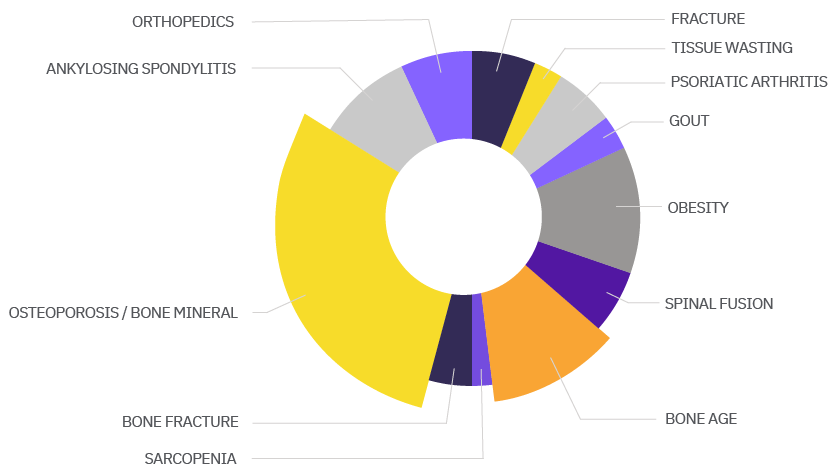
Calyx’s expertise spans the entire spectrum of MSK disorders. Click below to learn about Calyx’s experience in these indications: