Overview
Calyx Medical Imaging is a leading imaging core lab with the experience and ability to provide a wide range of services involving neuroimaging in clinical trials. Calyx Medical Imaging’s CNS group understands the unique and custom requirements required of Multiple Sclerosis trials and have the flexibility, creativity, bandwidth, and understanding already in place to effectively manage and support these clinical trials.
Case Study
A significant portion of our CNS experience is in Multiple Sclerosis (MS). For MS studies, we recommend conventional MRI sequences (T1, T1 post gadolinium, FLAIR and T2) for MS lesion evaluation (lesion counts and lesion volume). From the T1 sequence brain volume assessments such as total brain volume/brain volume change, grey matter volume, white matter volume, and CSF (cerebrospinal fluid) volume can be derived. Calyx can support further exploratory measures analyzed on Magnetization Transfer Imaging, Diffusion Tensor Imaging and Double Inversion Recovery. As a basis of a robust independent review assessment, standardized image acquisition is key. Thus, site training and imaging acquisition guidelines are critical for consistent and high-quality MRI images.
Case Study
Calyx Medical Imaging played a vital role in a phase 3, double blind, randomized, multicenter trial in patients with relapsing-remitting multiple sclerosis. Based on the centrally assessed imaging data it was shown that the investigational drug significantly reduces MS lesion activity. The superiority of the investigational drug against interferon treatment was also demonstrated on clinical grounds (relapse rate reduction of 52%). The trial was the largest phase 3 clinical trial program ever submitted to the FDA for a new MS drug at the time.
Example of MS lesion count and volume assessment.
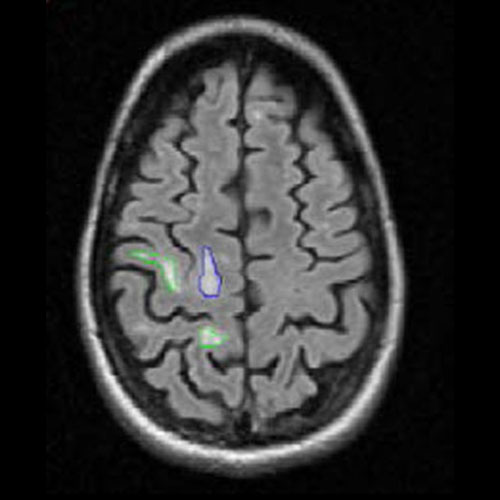
FLAIR image with lesion count and volume of new (green ROI) and enlarging (blue ROI) T2 hyperintense lesions.
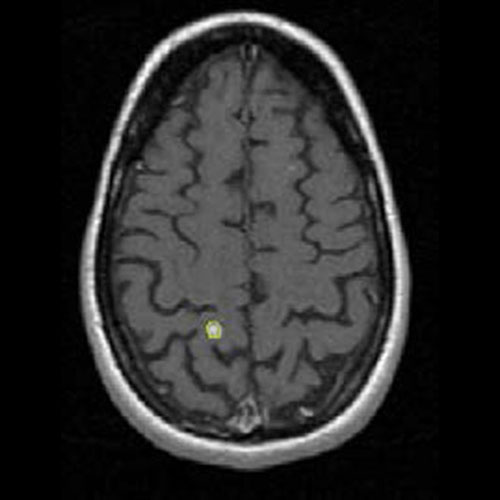
T1 Gadolinium enhanced sequence with lesion count and volume of T1 hyperintense lesions (yellow ROI).
Experience
Calyx Medical Imaging’s experience is drawn from managing over 2600 trials to date which include more than 4.4 million images from roughly 155,000 sites globally. Within this experience is our management of nearly 180 CNS protocols and over 30 MS studies.