Since the late 1990s, the number of countries participating in clinical trials has increased by 78%,[1] but this global expansion has not increased the diversity among participating patients. At the same time, clinical trial sponsors face patient enrollment challenges — particularly in oncology research, which is growing rapidly and increasing in complexity of study design and eligibility requirements.[2] As a result, finding and competing for the right patients is a hurdle that needs to be addressed.
A solution to this as an industry is in improving clinical trial patient diversification. This includes service providers, including centralized medical imaging providers, who must challenge their processes to support the enrollment of a more diverse patient population.

“Our system is somewhat biased toward participants who can move around easily, which doesn’t support the scalability we need in patient access to close the diversity gap.”
– Lindsey Kane, VP,
Technology Solutions – Medical Imaging, Calyx
Digging Deeper
Prostate cancer affects Black males ~2X more than White males.[3] Thus, the current situation of Black males only constituting 3% of PCa trial enrollment is misaligned with the disease manifestation in the general population.[4] Likewise, while the incidence of breast cancer is relatively equivalent between Black and White Americans,[3] White women make up 78% of breast cancer trial participants and Black women only 2.5%.[4]
The impact of these discrepancies is threefold: it produces data that does not reflect the general population, it increases regulatory risks to product labeling based on a therapy’s safety and efficacy in a specific demographic, and it limits ethnic minorities’ access to potentially impactful experimental treatments.
Expanding Site Access
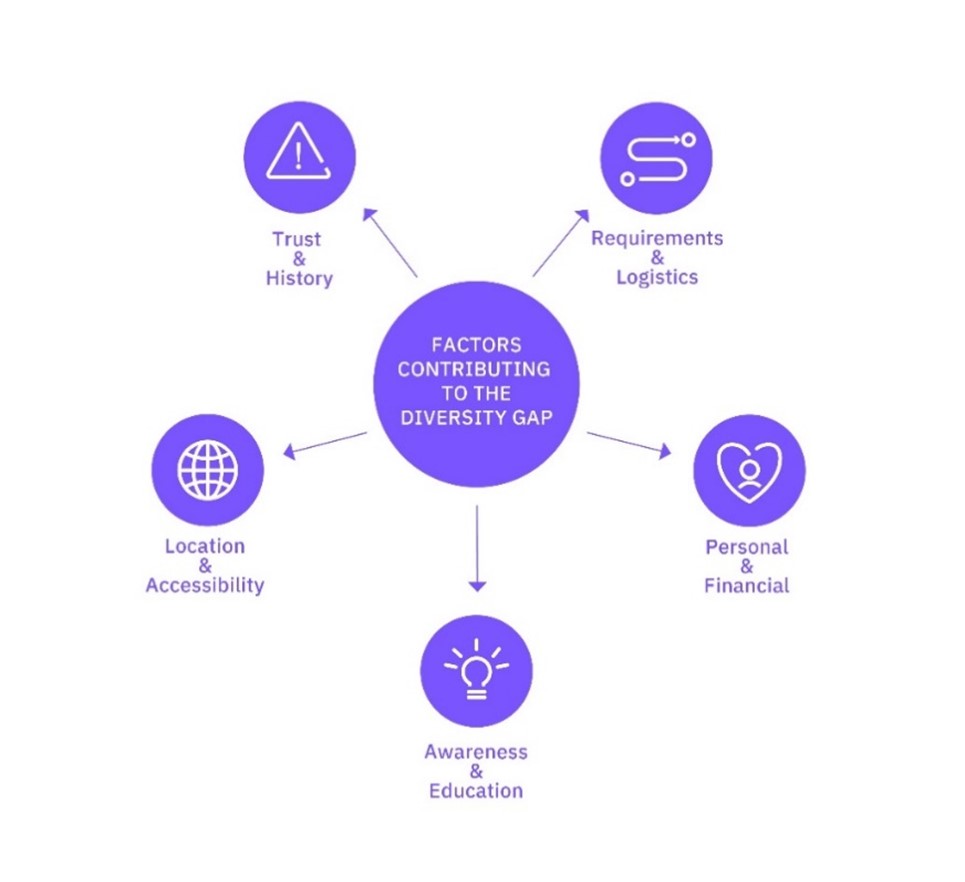
Among the factors contributing to the diversification gap (figure 1) is investigative site location. Currently, large academic medical centers make up a high concentration of clinical research sites, limiting access to the broader population that may be served at local community hospitals. Additionally, since participating in a clinical trial requires frequent and logistically challenging investigative site visits, many patients choose to not participate due to the need to take time off from work, secure childcare, and incur the cost of travelling.
This begs the question: If we limit site participation based solely on existing relationships, are we limiting our access to patients?
How Can Central Imaging Help?
In clinical trials that require medical imaging, every image counts — especially when imaging data is tied to a primary or secondary endpoint, or to safety or exclusionary criteria.
By leveraging an imaging core lab’s site database and by partnering with enterprise image exchange providers, we can expand options for image acquisition beyond the academic medical centers. In fact, one PI could have multiple centers associated with their investigator site to reduce the burden of patient travel. Traditionally, imaging core labs have been open to qualifying multiple imaging facilities within a trial. The next step is to proactively plan the identification of more accessible centers that will support patient diversification.
Another area of improvement is providing access to zero–technology footprint solutions, which will enable ease of site onboarding, simplify electronic image transfers, and support sites with the execution of local clinical research imaging reads. With fast image-upload and onboarding times, and by making imaging guidelines and trainings available online, we can help expand site participation while maintaining quality and consistency.
By identifying more imaging facilities and providing simplified processes, pharmaceutical companies and CROs can create modified feasibility plans and expand their network of sites, ultimately increasing their reach to a more diverse patient population.
Conclusion
Ultimately, we know we have a systemic problem with patient enrollment, and diversification is part of the solution. And that our current site selection approach contributes by biasing toward participants with access to major centers and who can travel easily and predictably. If we all do our part in tweaking our processes and services, we can overcome this challenge and better achieve our goal of gaining full knowledge in order to develop the safest and most effective treatments and driving human health evolution with velocity.
References
[1] When will clinical trials finally reflect diversity? (nature.com)
[3] CDC, 2019
—Incidence stat: USCS Data Visualizations – CDC
—Enrollment rates stat: Men of Color Remain Underrepresented in Prostate Cancer Clinical Trials – The ASCO Post
[4] FDA, 2019