Situation
A leading biotechnology company transforming their business across multiple therapeutic areas came to Calyx for help in designing an IRT system to overcome numerous obstacles:
- Some compounds were owned jointly with other companies, requiring more flexibility and adaptability in their IRT designs
- Delayed protocol feedback from regulatory agencies increased pressure on study timelines
- Rapidly expanding pipeline resulted in increased IRT deployment demand in support of more trials
- Already strained resources were expected to roll out studies faster and in parallel to keep up with the portfolio growth
- Other system-wide impact items included protocol amendments, regulatory reporting, and response capability
- Increased demand for more trials taxed their internal resourcing and capacity
Challenges
Calyx was asked to come up with a solution that would alleviate the RTSM-related pressures on the study team, freeing them to focus on other critical development needs. The challenges put forward for the RTSM technology were:
- Create repeatability in the design and delivery of the Interactive Response Technology (IRT)
- Streamline processes such as communication channels and study requirements documentation
- Create efficiencies for sites and in the delivery of each study’s IRT implementation
- Quickly implement protocol amendments to address the lag time of regulatory response
The expectation was that when these key areas were addressed, the biotechnology company would have the agility and flexibility needed to conduct their trials with confidence, ensuring continued support and growth of their portfolio. Naturally, this had to be accomplished without introducing risk or loss of data quality and accuracy.
Additional expected outcomes included the ability to:
- Ensure true alignment between IRT delivery and study teams
- Increase access and visibility into patient and supply chain data
- Leverage automation as part of the solution
- Increase efficiency in end-to-end IRT delivery
Reduce if not eliminate:
- Time needed to gather, document, and review requirements
- Need for custom programming
- Time required for User Acceptance Testing
Solution
The IRT system Calyx implemented was a business solution, encompassing both technology and service and ensuring that the people, technology, and processes were agreed upon by both organizations. The technology part of the solution included the development of IRT standards as well as the supporting business processes. It also required some processes to be adapted or new ones created.
Standards
Developing IRT standards was a collaborative process, which ensured the standards covered the three typical levels of functionalities in the IRT system:
- Core standards
- Sponsor standards
- Protocol requirements
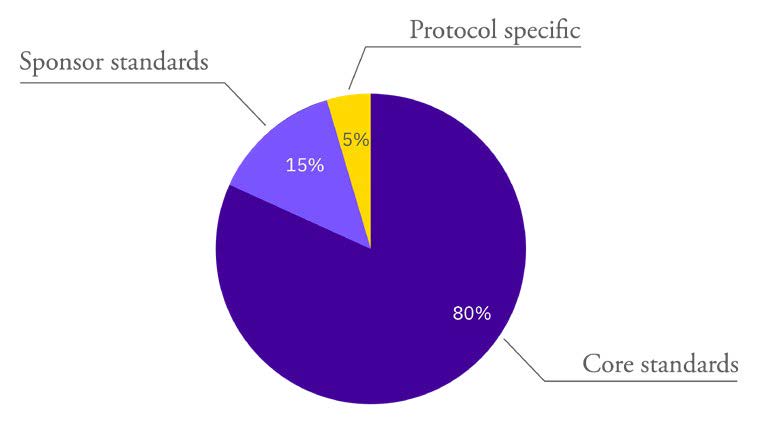
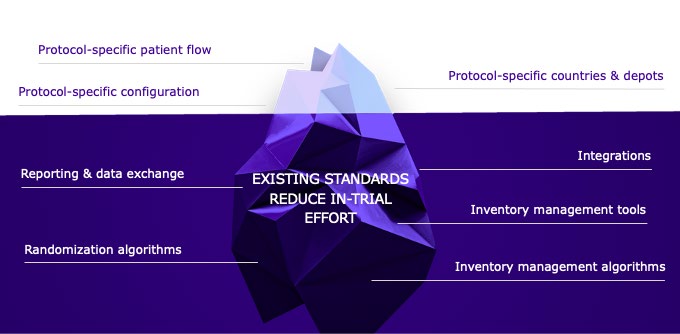
Typical functionalities of Calyx’s core IRT standards address such items as randomization algorithms, inventory management functionalities and algorithms, drug accountability workflows, and integrations. As a result, Calyx standards come with inherent system flexibility and adaptability to align each IRT deployment to the study protocol without the need for custom programming.
Sponsor-level standards encompassed their commonly used functionalities or settings, such as countries, depots, and inventory management preferences, all linked to their existing processes.
Process-related items included how project communication or standards adaptation would be handled, the communication channel, forms, templates, and other standard documentation such as project agreements, unblinding addendums, and project requirements form. They also included site-level processes identifying how site users were to interact with and utilize the system. This drove some of the biotechnology company’s specific user access requirements and standards development through reporting accessibility and useability.
Partnerships
As part of the solution, both organizations formed a partnership which included aligning the Calyx IRT delivery team with the client’s study team by therapeutic area. Portfolio directors, solution designers, engineers, validation, and support personnel, along with commercial business development and solution consultants were aligned as part of the partnership.
Portfolio and business development directors attended monthly governance meetings where the pipeline was reviewed, and high-level planning and agreements were made from a pipeline support perspective. Additionally, discussions included any items that may have had an impact on the solution such as standards enhancements or changes required to align the standards to business changes or to simply drive new requirements.
Benefits
- Increased efficiency in design, delivery, and communication, while reducing risk and providing the visibility and traceability requested
- The sponsor engages with the same IRT delivery teams across multiple trials, building trust and mutual understanding
- Increased site and staff efficiencies provide the ability to support sustained growth
- Increased data quality and consistency through standards and automation
Results
- Sponsor realized the value and benefits of program management
- Therapeutic area team alignment means studies are delivered by same team over and over
- Ability to adapt to un-expected life altering events: COVID Program successfully supported with 10 study go-lives in record setting deployment timelines
- Demonstration of how program management approach combined with client standards and processes through a true partnership increases efficiencies and sets all up for future successes
One of the truly measurable results of the combined standards and partnership solution is the enhanced efficiency and adaptability the biotechnology company experienced while responding to the COVID-19 pandemic. Based on a solid partnership between the therapeutically aligned Calyx IRT team and the COVID-19 study team, the IRT system was delivered within six days, freeing the sponsor’s subject matter experts to focus their efforts on solving issues linked to the effect of COVID-19 on other ongoing trials.
The biotechnology company was granted emergency authorization of its compound and was able to make one of the first COVID-19 treatments available to patients and first responders. Since the pandemic started, the biotechnology company has deployed ten COVID-19 studies of potential COVID-19 treatments, all initiated within similar expedited timelines.
The sponsor knew exactly what could be achieved with this very important program. Thanks to a strong partnership built over the years, they knew they could trust Calyx to deploy their program of COVID-19 studies accurately and in record time.
Key Highlights
- Ability to support any type of randomization
- Statistical designers with average 7+ years’ experience in IRT and 250+ adaptive trial designs delivered
- Secure data blinding, resulting in safe navigation through Calyx IRT and safe communication with our helpdesk
- As little as 4 weeks from requirements approval to UAT
- Integrations with eClinical suites (EDC, ePRO, central labs, supply management, etc.); Calyx IRT successfully exchanges more than 10,000 files with other systems daily
- IRT-dedicated helpdesk, specialized in managing the challenges of patient and inventory management for your sites and trial teams
- IRT self-service tools allow you to quickly adapt to varying site recruitment levels, change/add supply strategies, and activate new countries and depots
















