We have talked on many occasions about how IRT can help sponsors reduce the cost of drug management, by applying clever settings that reduce the amount of drug shipped to sites. One of those advanced IRT settings enables the management of pooled medication across trials that use the same compound(s).
Medication pooling is possible when more than one protocol operating at the same depot and/or clinical sites uses the same medication. Not only is it a good way to reduce medication wastage, but it is also an effective solution to address availability (scarcity) concerns or other restrictions that affect the supply of medication. When considering sharing medication across trials, sponsors have the option to apply medication pooling at depot level or at site level.

“Medication pooling is a good way to reduce medication wastage and to address availability concerns that affect the supply of medication.”
— Sylvain Berthelot, Technical Solutions Director, Calyx
Depot vs site level pooling
Depot and site-level pooling have different supply chain and IRT setup considerations. The level where the pooling takes place will impact on kit labelling, depot supply management, and site supply management. Both levels also provide different savings potentials, site pooling offering the highest medication savings.
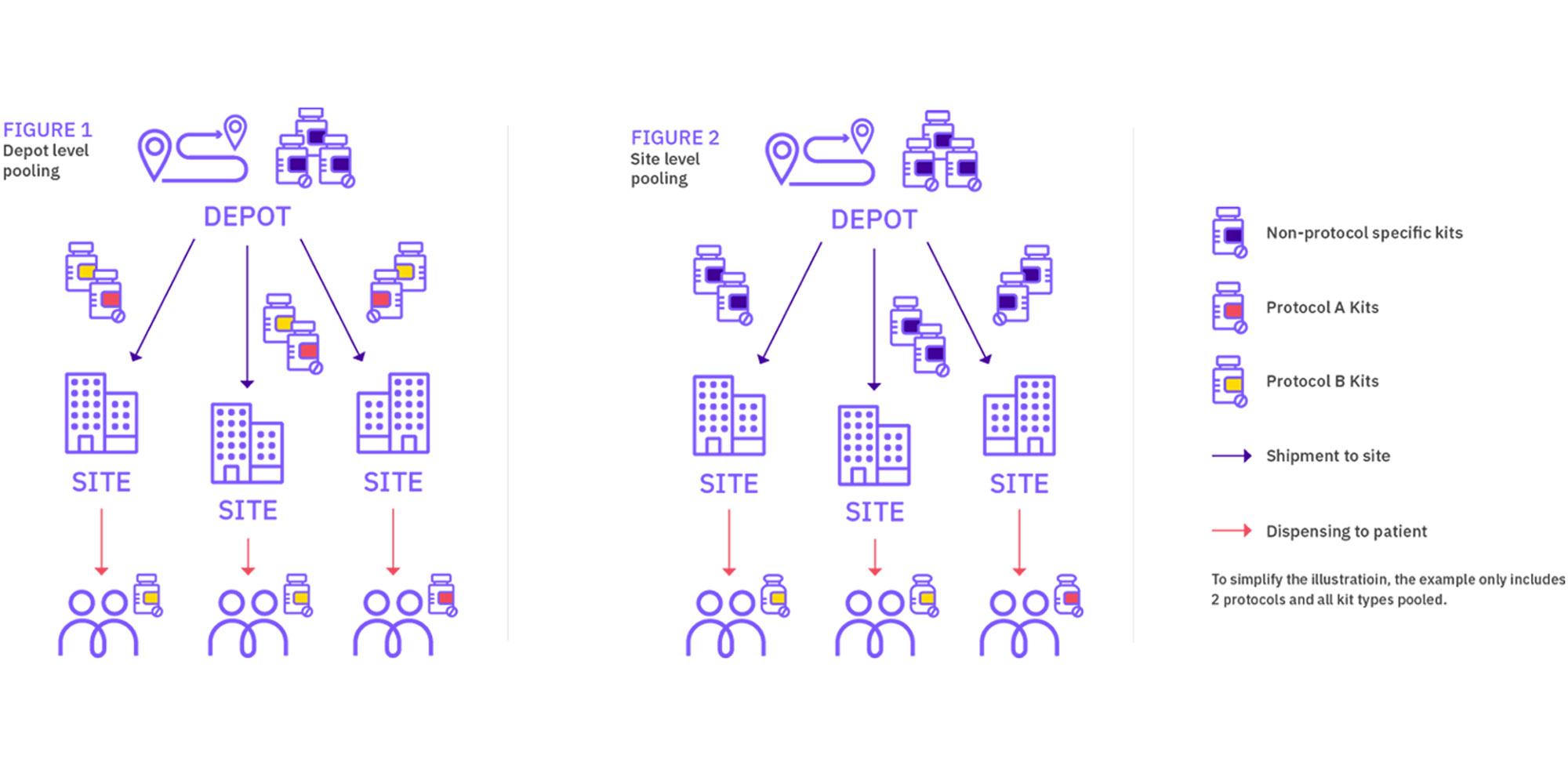
In simple terms, depot medication pooling consists of managing medication across trials at the depot but having protocol-specific medication at the site (even if the kit types are identical). On the other hand, site medication pooling results in managing medication across trials both at depot and site levels. The latter solution is always felt to be the best approach, however determining the size of the one pooled stock for all events in the group of studies could be complex.
How does IRT help?
Considering that it controls what medication is shipped from depot to sites, IRT plays a crucial part in the setup of medication pooling. In fact, IRT is one of the key enablers of medication pooling: it allows the use of the same medication across multiple trials, and it ensures each single trial “sees” the same pooled stock quantities, statuses, and locations. Maintaining those records accurately and in real-time is crucial to avoiding conflicting information and orders between trials. As such, IRT is a safeguard preventing issues in a medication pooling context.
Clinical supply managers will be wondering how supply strategies are managed in IRT. It will depend on the type of pooling applied for the program:
Depot-level pooling
When the IRT generates a new shipment request for a certain site, the medication selected to fulfil that shipment request is not pooled anymore, it becomes protocol-specific. This means that site supply strategies are applied at each trial independently and the IRT does not consider if the site already has medication of the same type that may have been shipped for another trial.
Site-level pooling
Shipment requests from depot to site take into account the needs across all the trials for which medication is pooled, so supply strategies should be built with this concept in mind. Supply strategies do not need to be identical across trials, although it may be useful to define the same strategy that works across the program. Our recommendation is to consult with an IRT expert to define the right strategy depending on the trial and/or program needs. With site-level pooling, adding a trial to the pooled program may require amending existing strategies for other trials in the program depending on how they have been setup.
In both scenarios, the resupply algorithm can still include patient-focused predictive supply to cover the needs of ongoing patients across the program.
The definition of supply strategies becomes more complex if protocols include both pooled medication and protocol-specific kit types, or if the sourcing strategy varies between kit types, with some medication being locally sourced, and others sourced centrally. Thanks to their extensive experience in clinical inventory management, Calyx supply management experts understand and apply the best practices that help sponsors adapt to any requirements.
Conclusion
Both depot level and site level pooling options can help reduce drug wastage and study-specific supply management oversight, the site level solution being the most effective. These solutions will not apply to all programs, as there is a need to have sufficient overlap in same kit type requirements across protocols for it to be beneficial.
Sponsors also need to consider the impact on labeling and should refer to regulations in force across all countries included in each trial to make sure they follow the right recommendations. Having multiple labels on kits can become confusing to depots and sites, specific labeling solutions are likely to be required to reduce both confusion and risk of error.
Looking to the future, solutions such as e-labeling should help support medication pooling strategies, as an integration between IRT and the e-label could automatically update the protocol at the right time, reducing depot and site burden, and increasing compliance.
















