The COVID-19 pandemic changed the conduct of clinical trials. Patients could not get to clinics for scheduled visits, so accommodations had to be made for home visits, telephone calls, and virtual visits via video. EDC study design needed to adapt to these new methods of clinical trial participation to continue capturing important trial data so trial sponsors and CROs could keep their studies on track.
Problems
Some of the EDC challenges that needed to be overcome included:
-
Tracking alternative methods of conducting visits
-
Enabling workflow to accommodate alternative visits
Solution
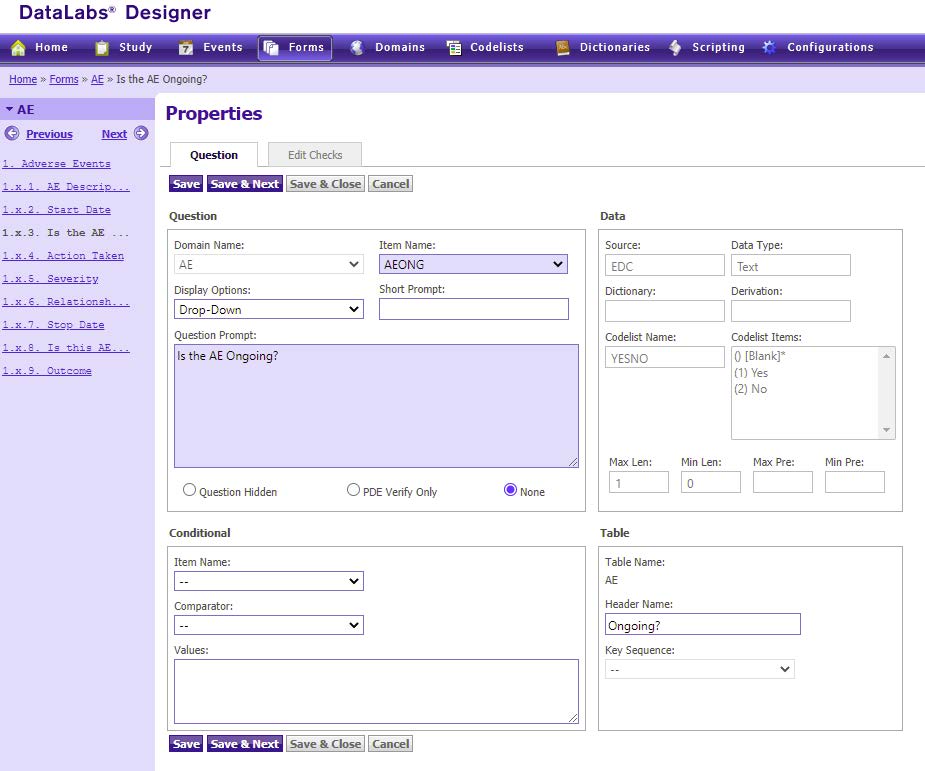
For one study, the CRO used Calyx’s EDC Design Tool to create a COVID Impact page which captured the details of how COVID impacted study participation. Questions were created to capture the occasions where a scheduled visit or treatment was missed or delayed due to a COVID related issue. If the visit was conducted as scheduled, a data point was created to capture whether it occurred via telephone, video, or a health practitioner’s visit to the patient’s home.
Depending on the method in which the visit took place, specific CRFs were triggered for data entry. For example, if the visit was conducted in the clinic, the Vital Signs and Physical Exam forms would publish, but not in the case of a telephone visit. If the visit was conducted via telephone, the telephone contact form would publish. Implementing these conditional forms simplified processes for site personnel, sparing them from having to identify forms that were not applicable and having to take the extra step of marking those CRFs not collected in the system.
To assist with workflow, queries were configured based on how the visit was conducted. For example, if the visit was conducted via telephone, a query was issued to remind the site to complete the Adverse Event and Concomitant Medication forms. These reminders prevented missing or incomplete data and forms.
These changes to the Calyx EDC configuration demonstrate the system’s flexibility in adapting to the needs of clinical trials in the COVID era.
Result
The flexibility of Calyx’s EDC system allowed the sponsor to adapt to a hybrid decentralized trial model, giving them traceability of how data points were collected to support the validity of the data.
Contact hello@calyx.ai to learn how Calyx’s EDC Design Tool gives you the flexibility to design and run trials more efficiently.
Efficient EDC Design
Effectively transforming a protocol into a full EDC study is of paramount importance in ensuring trial success. Without a robust, user-friendly design tool, the process of building EDC studies becomes protracted and inefficient. The problem is often exacerbated by the ever-growing complexity of study designs and widely dispersed locations of study design teams. It is therefore critical for study teams to be able to design in a collaborative environment using tools that promote standards and best practices.
The Calyx EDC design tool provides web-based study design, allowing users to efficiently build Calyx EDC studies through an intuitive interface, which guides you through each aspect of the design.
Users can streamline study creation by choosing design components from a centralized repository of existing studies, which facilitates the enforcement of design standards. The robust toolset makes it easy to configure CRFs, coding dictionaries, code lists, derivations and edit checks.

KEY FEATURES
- Centralized storage and management of study designs
- Promotes and supports CDISC standards for eCRF design
- Leverage our library of CDISC compliant forms and code lists
- Import any CDISC ODM study design
- Flexible event scheduling accommodates multiple cohorts and treatment cycles
- Dynamic forms and events facilitate complex workflows
- Over 30 keyboard shortcuts, i.e., hotkeys
KEY BENEFITS
- Intuitive web interface makes study design simple and efficient without the need for advanced programming skills
- Streamlined study build process and workflow to reduce development cycle time
- Global and collaborative study development enabled by web-based access
- Ability to develop, implement and enforce organization-wide eCRF design and data standards
- Efficient study design utilizing validated study components
- Delivered as a cloud solution, eliminating the overhead of system investment and maintenance issues
Endless Possibilities (Nearly)
Benefits of the Calyx design tool for visit structure include:
- Dynamic Events and Forms allow for visits and included CRFs to be tailored to each patient
- Repeating Events facilitate studies where the ultimate number of visits for a subject is uncertain
- Multiple Event Schedules provide for customized visit schedules based on treatment group or study arm
Because the Calyx EDC design tool is cloud-based, users can access their studies from any computer, anywhere in the world.
The Calyx design tool manages all study builds for our proven EDC Solution. Features renowned for intelligently streamlining workflows include:
- Product convergence with Calyx IRT for randomization and trial supply management, allowing site users to VIEW randomization and medications dispensed in real-time directly from Calyx EDC
- Flexible data model allowing real-time access to view and retrieve data
- Web services enabling interoperability with any third-party applications
Proven. Flexible. Easy.
Since 2002, Calyx EDC has helped global biopharmaceutical sponsors, clinical research organizations (CROs), and medical device manufacturers realize the true potential of EDC technology. Calyx EDC is synonymous with exceptional usability and rich functionality, underpinned by renowned performance, scalability, and resilience.
Uniquely focused on simplifying workflows and delivering superior user experiences, Calyx’s advanced EDC features streamline the entire process starting from study design all the way to collection, management, and reporting of clinical trial data.
Delivered through the cloud and backed by the full power of our leading eClinical portfolio, Calyx EDC offers total flexibility and ease-of-use for researchers of all types and sizes.
Datasheet

KEY FEATURES
- Targeted Source Data Verification (SDV) focuses resources where they are most needed
- Study design comparison eliminates manual comparisons and reduces validation time required to review design changes
- Robust safety reporting provides real-time notification of critical safety events that are time-sensitive for processing
KEY BENEFITS
- Leverage Targeted SDV to reduce on-site monitoring activities and the associated costs
- Make data-driven decisions to focus site monitoring priorities
- Simple user interface enables ease of use and rapid study build
- Improve patient safety and data quality
- API web services with event-driven architecture allow you to integrate with a variety of systems or devices
- Patient profiles available on-demand to facilitate cohort or safety review
- Normalized local lab values standardize results for analysis
- Identify efficiencies in study design (comparison tool)
- Increase data availability and visibility (APIs)
- Accelerate data review (patient profiles, labs)
Study Design
Effectively transforming a protocol into a comprehensive set of eCRFs is critical for EDC deployment. The Calyx design tool is an innovative study build application that provides study teams with the tools necessary to build an electronic study, all in a truly collaborative environment. Together with the expertise of our professional services group, the Calyx design tool ensures efficient and optimal study build processes.
Highlights
Intuitive, web-based design tool
- Simple drag-and-drop features to configure CRFs, coding dictionaries, code lists, derivations, and built-in edit checks
- Centralized repository of study design objects and metadata
- Import previous study designs using XML
- Create form libraries by reusing components from existing studies
- Visual comparison of study design changes between studies
- No advanced programming skills are required. Point and click conditional fields configuration
Data Collection
Calyx EDC is designed to provide the best user experiences possible with the core features you expect, delivered in an easy-to-use interface. Built with utmost usability in mind for collection of study data, the easy-to-navigate interface and workflow-driven display of permissible actions facilitate highly streamlined user workflows for site activities. Renowned for its ease of use, Calyx EDC’s user interface empowers investigative sites to effectively utilize EDC with a minimal learning curve.
Highlights
Intuitive, web-enabled interface
- Built-in edit checks to ensure complete and valid entries
- Clear visibility of unscheduled visits in proper sequential order
- Robust eLearning features for site training and certification
Role- and workflow-based views and displays
- Action palette facilitating efficient user workflows
- Display of only applicable visits for individual patients
Integrate data from central labs, imaging, ePRO and other third-party systems
- Robust APIs provide complete visibility and audit trails of integrated data
- Eliminate data discrepancies, minimizing queries and reconciliations
- Store all data within a single system, eliminating the need to merge multiple databases
Study Management
Managing today’s clinical trials is a complex and challenging task, involving many different people, processes, and systems. Study teams need to respond to rapidly changing situations and make timely, informed decisions throughout. Calyx EDC has a proven track record of helping organizations optimize valuable resources, gain efficiencies, and ensure quality data.
Highlights
Advanced suite of data management capabilities
- Extensive search and filter options using multicriteria selections
- Flexible CRF flag options for full range of review types
- Customized event-driven notifications and alerts for collaboration across multiple users
Dynamic generation of forms and visits enable collection of targeted and relevant data
- Ability to tailor and generate specific CRFs for each individual patient
- Optimal, intuitive data entry experiences for site users
Comprehensive reporting capabilities
- Dashboard with visual displays of EDC metrics to aid strategic decision making
- Full range of standard reports and flexible ad-hoc reporting
- Drag and drop interface without the need for advanced programming
Advanced data export capabilities accelerate analysis and decision making
- Ad hoc access and review of data
- Export in SAS transport and ASCII Delimited files, available on-demand or at scheduled intervals
- Clear visibility of unscheduled visits in proper sequential order in exported data
Powered by Leading Technology Infrastructure
Calyx EDC is built to provide ultimate performance, quality, and availability through intelligent architecture and best practice capacity provisions. The federated architecture of Calyx EDC and the resulting scalability enables us to support studies of all sizes, including extremely large-scale clinical trials.

Calyx EDC Professional Services
- In-depth study design and consulting services
- Development of flexible, customized training curriculum tailored to individual customer requirements
- In-person and virtual training programs
- Built-in, self-directed eLearning
- Consulting, guidance, and support to achieve alignment with client objectives
Customer Care Services
- Supports all leading eClinical technologies
- Quick turnaround times
- Proactive high-quality support
- Services tailored to client requirements
- 250+ local languages
- Single point of contact 24/7/365