This blog was written in February 2023. Please note that due to the fast-moving nature of this topic, dates are subject to change.
In January 2023, the ICH released updated Regional Implementation Information for eCTD 4.0. This is set to be a crucial year in the implementation of eCTD 4.0 with technical pilots running in a number of regions, and the FDA beginning to accept eCTD 4.0 submissions on a voluntary basis.
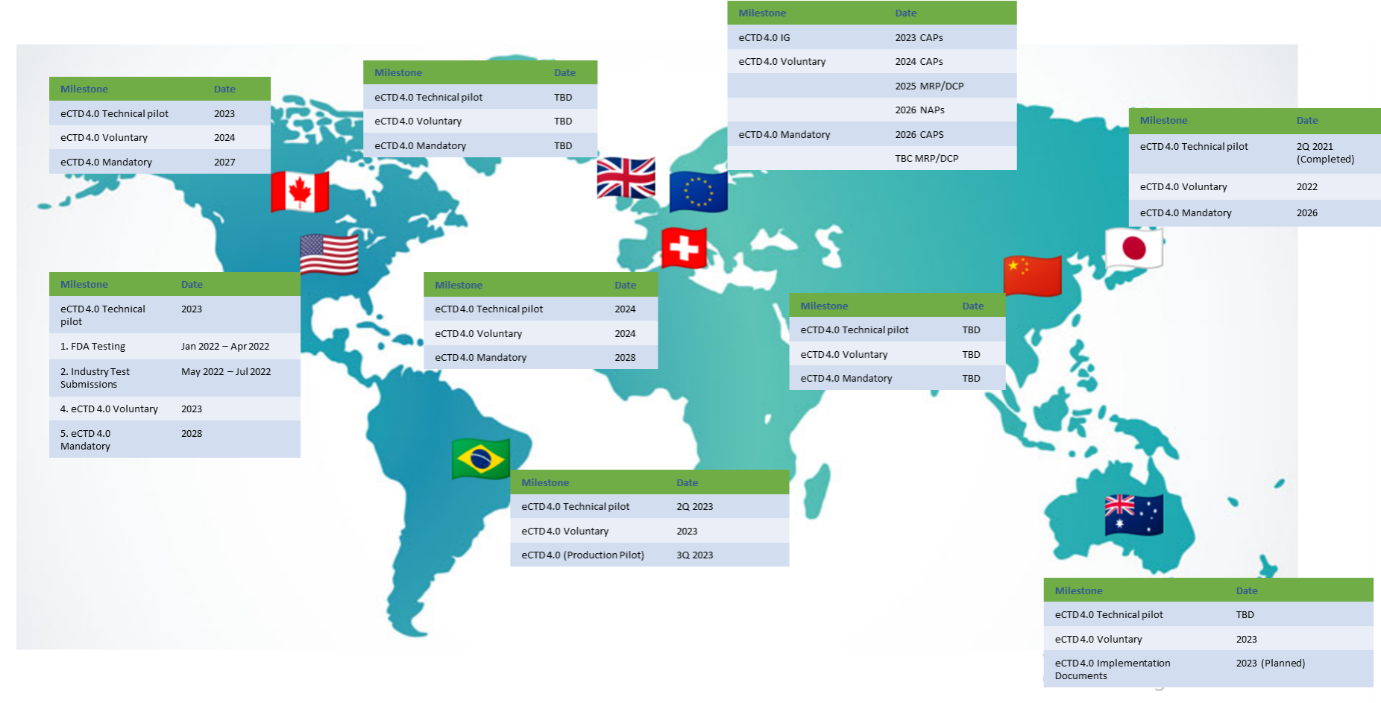
Let’s start with the FDA, which released the eCTD v4.0 Module 1 Implementation Package in September 2022. The IQ pilot began in June 2022 and runs until the end of March 2023. In 2023, CDER and CBER intend to begin accepting new eCTD v4.0 applications on a voluntary basis. Subsequent phases will cover v3.2.2 applications and two-way communication, and eCTD 4.0 will be mandatory starting in 2028. Implementation planning documents are available at fda.gov.
PMDA completed their pilot test in the second quarter of 2021 and began accepting applications in v4.0 in the year 2022. The transition phase will last through 2026 at which point eCTD v4.0 submissions will become mandatory in Japan.
Early in 2023, EMA intends to release the Implementation Guide and associated standards guidance. Timelines for voluntary and mandatory submissions are staggered based on procedure type: voluntary submissions for CAPs start in 2024 and become mandatory in 2026, while voluntary submissions for MRP/DCP and NAPs start in 2025 and 2026 respectively. The mandatory use dates for MRPs/DCPs have yet to be confirmed by the EMA. Updates are available on the EMA’s eSubmission site.

Source: EMA EUROPA
The next milestones for Health Canada have been developed based on stakeholder feedback and work with ICH partners and other regulatory authorities on the international deployment strategy for eCTD v4.0. Health Canada are planning to complete pilot testing in 2023, accept voluntary submissions in the year 2024, and will make eCTD 4.0 submissions mandatory from 2027. The guidance document, “Preparation of Regulatory Activities in eCTD Format’ will be updated to reflect the new specifications once the choice to implement eCTD v4.0 has been made. Draft guidance has been released and is available on Health Canada’s website.
Swissmedic held a public hearing on the Swiss Regional Implementation Guide back in 2020, but the final publication of the guide has yet to be made available. Meanwhile, Swissmedic plans to run a technical pilot in 2024, followed by voluntary submissions in eCTD 4.0 format the same year. Swissmedic anticipates mandating the use of 4.0 by the year 2028.
The voluntary implementation deadlines for ANVISA and TGA are 2023; a technical pilot for ANVISA is scheduled for the second quarter of 2023, while a decision is still to be made for TGA.
The MHRA, ECOWAS, and ECAs regions do not currently have any plans to deploy eCTD v4.0; they will follow suit once other regions have paved the way.
The next five years will see rapid change as national authorities complete technical pilots and begin accepting eCTD 4.0 submissions.
Learn how Calyx RIM helps you maintain compliance with all regulatory requirements.
















