Considerations for Life Sciences and Vendors
In our previous blogs we discussed the people and process considerations as well as the impacts of new concepts, terminologies, and the increased reliance on structured data during an eCTD 4.0 implementation. In this final blog in the series, we review some of the challenges for implementation and longer-term adoption both from a life sciences and vendor perspective
Timelines
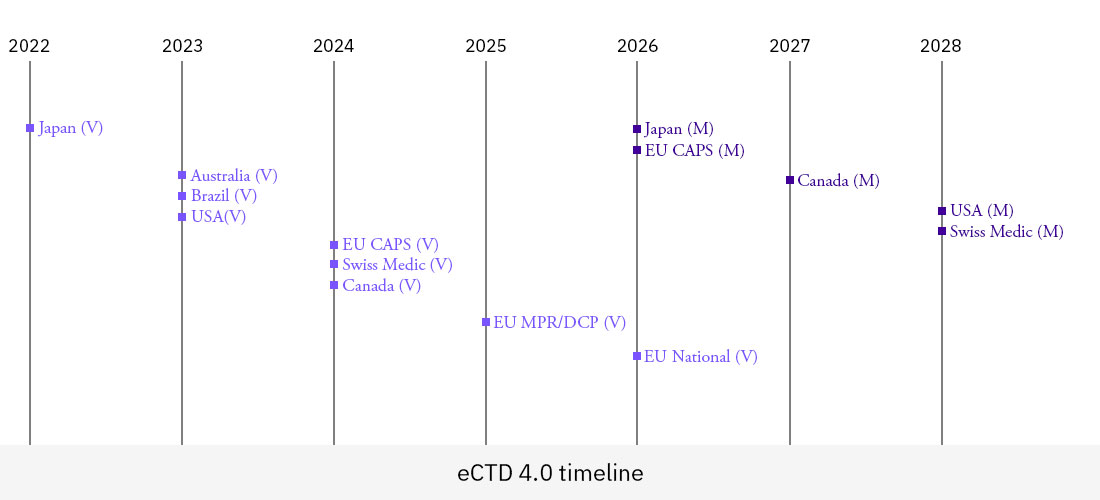
The timelines for implementing eCTD 4.0 across the different markets and regions is varied and subject to change. In the July 2022 ICH updates Canada moved the dates for voluntary implementation from 2023 to 2024 and for final implementation from 2026 to 2027. These timeline changes mean that it is often difficult for pharma companies to effectively plan their implementation; having to take a risk-based approach in assigning resources and prioritization. Additionally, the long lead times between voluntary and mandatory submissions coupled with the need to comply with other future regulatory requirements such as IDMP & PQ CMC make it difficult to prioritize and plan for eCTD 4.0.
For several in-scope countries e.g., Brazil and Australia and for Non Centrally approved products in the European Union, timelines for mandatory implementation are not yet known, which further complicate plans to move forward. While in some cases countries such as Thailand, South Africa and the Gulf Cooperation Council region have not provided any plan for their 4.0 implementation. In summary the implementation of eCTD 4.0 looks to be a protracted process which will continue to evolve way into the 2030s.

“Long lead times between voluntary and mandatory submissions coupled with the need to comply with other future regulatory requirements make it difficult to prioritize and plan for eCTD 4.0.”
– Karen Harry, RIM Expert
Below is a representation of the current timelines for voluntary and mandatory timelines (correct as of July 2022).
Transition
Another important area of discussion will be in deciding the plan for transition. As referenced above there are lengthy timelines from optional submission to mandatory submissions and there will need to be capabilities to manage a single application in both eCTD 3.3.2 and 4.0
This will require some very detailed planning and co-ordination with submission planners, publishers and authors alike. It will clearly be critical to have a well-defined plan for transition; a couple of considerations in helping to inform these decisions could include the prioritization of applications where there are minimal updates planned.
Working in close collaboration with submission planners, especially in larger organizations must be a priority, having access to the submission schedule and visibility of any variations to it will be critical.
Another obvious option to facilitate the transition would be to identify a subset of publishers with eCTD 4.0 subject matter expertise to manage the follow up submissions post transition. It would also make sense to target any new MAAs as candidates for eCTD 4.0 from the beginning of their lifecycle.
Vendors
Vendors will clearly be pivotal in providing eCTD 4.0 capabilities in their publishing solutions and in supporting future submissions in the new format while still maintaining support for current eCTD specifications, NEES, and paper publishing. New assembly templates will need to be provided to support the new 4.0 structure and updates to DTDs will also be required.
Validation tools will need to be updated to ensure that all criteria including the rules relating to the association of controlled vocabularies and sender defined keywords are included. Vendors will also need to focus on viewing tools, ensuring that the capabilities to review both the content and the structure of the eCTD 4.0 format are provided.
Other requirements that will need to be satisfied include the need to link to multiple controlled values and for those companies that have an integrated registrations and publishing utilization of the same list of values for both IDMP and publishing will be hugely beneficial, along with any future initiatives requiring those same values.
As with other ongoing initiatives there will be a reliance on FHIR (Fast Health Interoperability Resources) to generate the single XML backbone.
Although out of scope for the initial submissions, in the future vendors will also need to consider the need to support two-way communication so that agency feedback can be included. This in itself will introduce changes to business process to ensure visibility to the regulatory affairs team who are typically in receipt of such communications.
Importantly, vendors will also be expected to collaborate with industry to support agency pilots prior to the formal implementation of eCTD 4.0. These pilots will be crucial to the success for both industry and health authorities alike.