RTP-based US headquarters established to meet pharmaceutical companies’ and CROs’ increasing needs for reliable eClinical and Regulatory solutions.
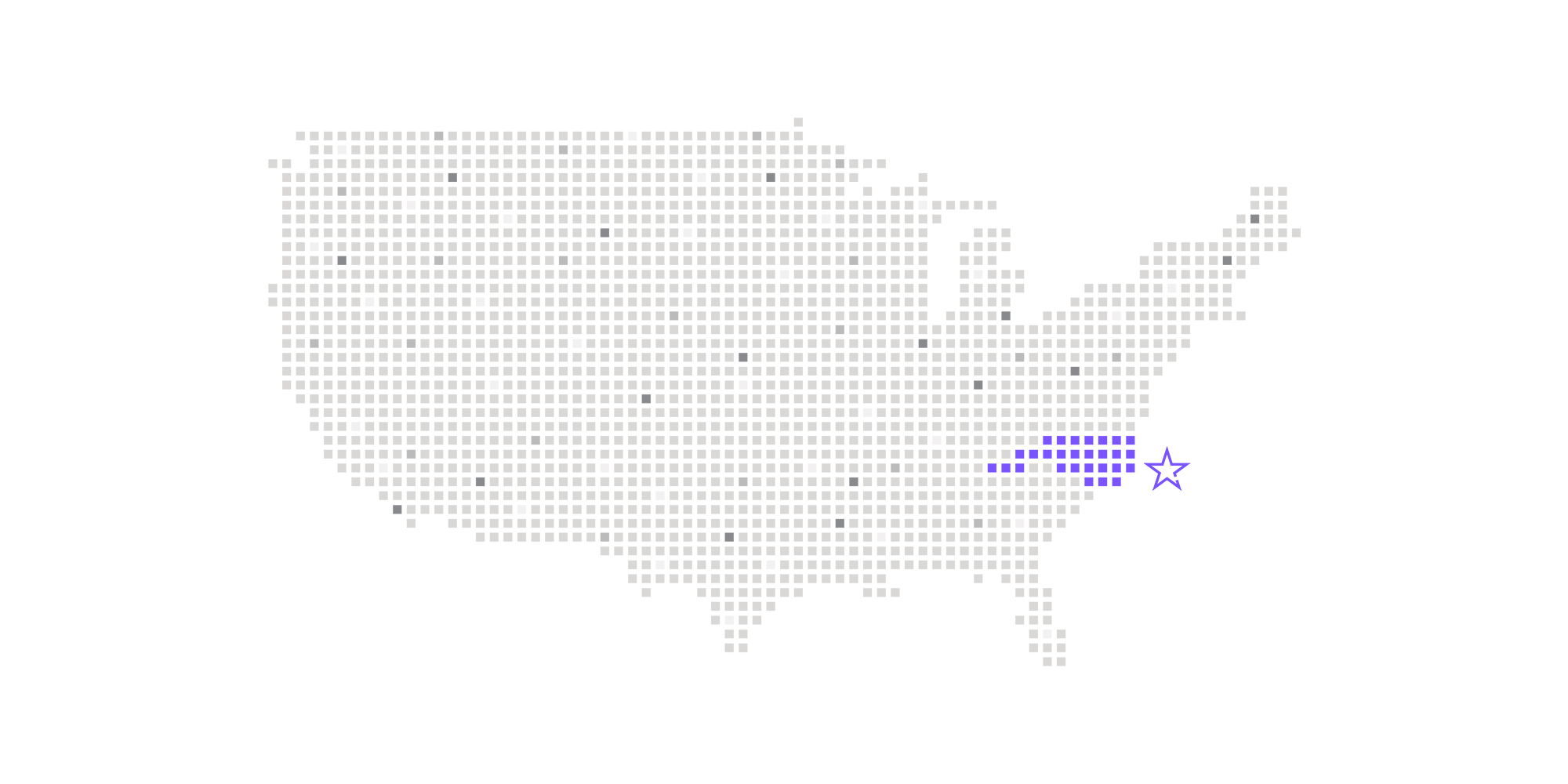
Nottingham, England and Morrisville, NC – July 19, 2021 – Calyx, the eClinical and Regulatory solutions and services provider most relied on for solving complex data challenges in clinical research, today announced it is expanding its presence with the opening of its US headquarters in Morrisville, North Carolina.
Adjacent to Research Triangle Park (RTP), the facility was selected for its proximity to many of Calyx’s clients, including some of the industry’s largest pharmaceutical companies and clinical research organizations (CROs).
“The opening of our US headquarters is representative of the high value we place on our customer relationships,” said Gavin Nichols, CEO of Calyx. “Working closely with these many successful clinical trial sponsors and CROs – and leveraging the rich pool of experienced industry talent in the area – enables us to continue our mission of providing the innovative technology solutions and services our customers need to speed the discovery of new treatments.”
About Calyx
Through innovative eClinical solutions and services, Calyx turns the uncertain into the reliable, helping bring new medical treatments to market faster. With deep expertise in clinical development and more than 25 years of supporting trial sponsors and clinical research organizations (CROs), Calyx harnesses that intelligence and activates its potential to solve complex problems, deliver fast insights, and get new drugs to market every day.
CTMS | EDC | IRT | Medical Imaging | RIM
Take your trials further, faster at calyx.ai, LinkedIn, Twitter, or Facebook.
Media Contact:
Christine Tobin | Christine.Tobin@calyx.ai |+1 412-628-8598